- Author: Niamh M Quinn
- Author: Roger A Baldwin
- Author: Carolyn Whitesell
Pocket gophers can cause significant damage to valuable turf, girdle trees, and chew irrigation lines. Their mounds can create tripping hazards and lead to erosion concerns when found on slopes. Luckily there are multiple successful management options to choose from when it comes to managing pocket gophers.
Do I have a pocket gopher?
Pocket gophers are small burrowing rodents that are often identified from the damage they cause rather than a sighting of the animal itself. Pocket gophers spend most of their time below the surface and while it is possible to see them above the ground, or peeping out of a burrow (Figure 1), the easiest way to determine the presence of pocket gophers is by the crescent-shaped mounds they leave behind on the surface from excavating their burrows. Pocket gopher mounds can be differentiated from mole mounds by their shape—mole mounds will be round whereas pocket gopher mounds have the distinctive crescent shape. Pocket gophers do not hibernate and are active year-round, so it is important to be vigilant when managing pocket gophers.
Management options
There are many management options for pocket gopher control and in California, significant research has been conducted to better utilize and examine the effectiveness of many of these tools. While there continues to be restrictions on some of the toxic options for pocket gopher management, there are still multiple non-chemical options to choose from that fit well into an IPM plan. Pocket gopher trapping has proven to be one of these very effective methods.
Trapping
Trapping is an excellent option for managing pocket gophers, even if you think you have a large population. In general, there is only one pocket gopher per tunnel system, so once you have captured one, you can move to the next system. Be aware that during the breeding season, there could be both a male and female in the burrow system, and after the female gives birth and the pups are dispersing, you can find the pups in the burrow system with reproducing females. In these scenarios, you might capture more than one pocket gopher per trap set.
Trapping takes longer when compared to toxic bait application or burrow fumigation with aluminum phosphide but it can be highly effective when done correctly.
Make sure you have the right tools when it comes to trapping pocket gophers. It is important to have sufficient traps for the population you are dealing with. Focus trapping efforts on fresh mounds, as older mounds are less likely to be active. If you are unsure what mounds are active, you can knock over all mounds and trap at the new mounds that appear. Pocket gophers may not mound every day, so trap over more than one day to be successful. It is worth noting that it can be difficult to determine individual burrow systems from each other. Some practice is required to better define which mounds are likely from the same individual. If a trap set is not successful after 1 or 2 days, move it to a new tunnel location.
Trap selection (Figure 2) and placement may be important when choosing trapping as your pocket gopher control method. In studies, the Gophinator trap caught larger pocket gophers at a higher rate than the Macacbee trap. In addition, these traps often require larger excavations in the soil. For areas like high value turf, these traps may not the best option. An alternative trap is one resembling the GopherHawk, which may cause less damage because they don't require as large of an excavation in the soil.
When you set a pincher-style trap like the Macabee or the Gophinator, or a box-type trap like the Black Box or Black Hole, it is important to locate the main run of the burrow system. To do this, take a long metal rod like a screwdriver and probe around the mound. You will need to stick your probe into the ground at depths of about 4 to 12 inches. When you find the tunnel, you will experience a sudden drop in resistance. This skill is difficult to acquire but will improve with practice. Once you have located the main run, you need to excavate an opening in the tunnel to allow for setting of the trap. You can use a hori-hori knife or a trowel to make this hole, which may need to be more or less extensive depending on which style trap you choose.
If you are using a pincher-style trap that is set inside the tunnel, make sure that the tunnel you are setting the trap in is straight. You can check this by putting your excavator tool into the tunnel and making sure that at least 6 inches of it fits into the tunnel. This way you know the tunnel doesn't turn; tunnels with turns often allow the pocket gopher to bypass the jaws when activating the trap, thereby resulting in triggered traps that miss the gopher.
Several attractants have been tested but they did not influence visitation or capture rates of pocket gophers to traps. Attractants also did not influence the gender of pocket gophers captured. There is no impact of human scent on the success of trapping pocket gophers.
Is blocking light by covering trap sets necessary when trapping for pocket gophers? Covering and uncovering pocket gopher trap sets is time consuming and does not result in a greater number of captures. However, if you are trapping in areas with high foot traffic, there may be some benefits to covering your trap sets. You can cover the traps with sod, landscape cloth, or something like cardboard or plywood to prevent people or pets from interfering with your trap sets. In general, it is recommended that you cover sets when using box traps, since pocket gophers will likely plug tunnels before hitting the trigger wire of these traps if you leave them uncovered.
It can be helpful to tie a flag to the traps so you can easily remember where they were set. This can also help you recover the trap if the gopher drags it away.
When you recover a dead pocket gopher, remove it from the trap, put the gopher back in the tunnel, and cover it up. You can also double bag the animal and place it in the trash. Always wear gloves when handling pocket gopher or any other wildlife carcasses.
Toxic baits
There are several options for pocket gopher management using toxic baits. It is important to ensure that bait is placed correctly in the tunnel either by using a probe with a bait applicator or by hand using a funnel and spoon. Always read and follow the label. Many toxic baits that are used to manage pocket gophers require a restricted materials permit. However, there may be exemptions for products applied for structural pest control, industrial use, and institutional use.
Strychnine is the most effective type of bait used for pocket gopher management. This toxicant is an acute rodenticide where a lethal dose can be acquired after a single feed. Pocket gophers can develop behavioral resistance to strychnine. This enables pocket gophers to consume what is normally more than the lethal amount by periodically eating sublethal amounts. Pocket gophers also can be physiologically tolerant to strychnine since after they ingest a series of higher doses of strychnine, they can tolerate increasingly higher doses of it. It is important to rotate strychnine in with other management tools to avoid this type of resistance.
Zinc phosphide is also an acute rodenticide that is lethal after a single feeding. Bait shyness or taste aversion can be associated with this management option. Because of this, zinc phosphide may not perform as well as strychnine.
First generation anticoagulant rodenticides, including chlorophacinone and diphacinone, are multiple-feeding anticoagulants that are less toxic than strychnine and zinc phosphide. Since these baits require multiple feedings over 3-5 days, it is important to make sure that there is a continuous supply of bait during your treatment period.
Fumigation
Aluminum phosphide is highly effective, especially in moist soils. However, aluminum phosphide is a highly restricted material. It must be applied by a certified applicator, or the application must be supervised by a qualified applicator. You must have a restricted materials permit, a written recommendation to apply on production or non-production agricultural sites, and a Notice of Intent (NOI) from your local Agricultural Commissioner. You are also required to have a Fumigation Management Plan. You are not permitted to apply this product within 100 feet of a potentially occupied structure. The only place on school grounds where it is permitted to apply this product is on athletic fields.
Gas cartridges are not effective because pocket gophers seal off their burrow when they detect the smoke.
Carbon monoxide producing machines and carbon dioxide are registered for use in California against burrowing rodents. Carbon monoxide producing machines are registered devices, while carbon dioxide used for rodent control is considered a registered pesticide.
Carbon monoxide devices generate carbon monoxide which fills the burrow system and asphyxiates the pocket gopher. These devices include the BurrowRx, Cheetah Rodent Controller, CO Jack, and Pressurized Exhaust Rodent Controller (PERC) Machine. Research has shown that the PERC machine can be moderately effective at managing pocket gopher populations. Its efficacy increases in moist soil conditions. Some of these devices are more suited for urban applications and some for larger scale production agriculture. You are not permitted to use a carbon monoxide pest control device within 100 feet of a structure inhabited by people or domestic animals, whether occupied or not.
Carbon dioxide gas is a pesticide that is registered for use on several sites that include production agriculture, non-production agricultural sites, and residential areas. There are no distance restrictions for the application of this pesticide. It is important to follow the label. Solid carbon dioxide (dry ice) is not registered for use on pocket gophers.
Natural predators
Vertebrate predators—including owls, snakes, cats, dogs, and coyotes—eat pocket gophers. However, they will rarely control all pocket gophers in an area. Predators rarely remove every prey animal but instead move on to hunt in areas with more prey. In addition, pocket gophers have defenses against predators. For example, pocket gophers can evade snakes in their burrows by pushing up an earthen plug to block the snake's advance. Relying solely on natural predators might not control pocket gophers to the desired level. Research has shown that pocket gophers appear to be an important prey animal for barn owls nesting in perennial crops, and thus barn owls may be able to provide some pest control services in those areas.
Exclusion
Exclusion can be difficult and expensive for pocket gopher management, but it may be justified if you are trying to protect individual or high-value landscape plants. You can use hardware cloth (1/2-3/4-inch mesh) buried at least 2 foot deep with an additional 6 inches of hardware cloth bent at a 90° angle. You should also extend fences at least 1 foot above the ground because pocket gophers may move above ground to access the planting you are trying to protect. There are wire baskets available to protect individual plants or bulbs from pocket gophers. These baskets can also be fashioned from chicken wire. Remember that it is important not to restrict the growth of the plant inside the basket, so ensure the wire basket is large enough to accommodate the adult plant's root structure.
For more information about pocket gopher management, see the UC IPM Pest Notes: Pocket Gophers.
[Originally featured in the Spring 2024 edition of the Green Bulletin Newsletter for structural and landscape pest professionals.]
- Author: Andrew M Sutherland
Baits Eliminate and Prevent Subterranean Termite Colonies

Subterranean termites (Family Rhinotermitidae) are considered the most serious wood-destroying pests in the world, causing an estimated $32 billion in global economic impact each year. California is home to both native and introduced subterranean termite species (Figure 1). Infestations of wooden structures are widespread and common. Pest control operators (PCOs) have conventionally applied liquid termiticides to control these pests, usually as soil drenches or injections around structures. These treatments may not always be effective, however, especially if good underground coverage is not achieved, if local termite pressure is very high, or if dealing with the invasive Formosan subterranean termite in Southern California. Furthermore, the active ingredients in most liquid termiticides are increasingly monitored by the State as environmental contaminants and may be subject to legal restrictions in the future.
Bait systems for subterranean termites (Figure 2), which employ slow-acting insecticides that kill worker termites by preventing successful molting, may represent effective alternatives to liquid treatments. Baits, deployed within stations installed in the ground or in line with aboveground shelter tubes, have gained popularity during recent decades and are now considered the primary subterranean termite control tactics in many parts of the world. Adoption of bait systems in California has lagged most other regions, however. Reasons PCOs in California have reported being reluctant to use bait systems include 1) time required to achieve control is too long, 2) little efficacy data in California, and 3) the regular monitoring of bait systems is too labor intensive or otherwise does not fit established business models.
Recently, the third “adoption barrier” may have become less important: new product label guidelines allow PCOs to extend inspection intervals up to 12 months and allow for baiting without the previously required monitoring phase (provided the target pest is confirmed at the site). Considering the regular revenue streams created by “controlled service agreements”, where PCOs contract with property owners to prevent and control pests over a long term, these newer labels should drive more widespread use.
Some observations and case studies indicate that, indeed, bait system adoption is now slowly increasing in California. To address the other two reported barriers (speed of control and efficacy), we secured funds from the state's Structural Pest Control Board to evaluate and demonstrate three different in-ground bait systems in the San Francisco Bay Area and the greater Los Angeles area.
Bait Efficacy
Our first objective was to evaluate efficacy at single-family homes. To do this, we collaborated with five different PCO companies who expressed interest in the new business models made possible by the newer bait product labeling guidelines. Some of these companies had experience with baits, while some gained their first experiences through this project. Companies received research stipends to subsidize their participation. Fifteen single-family homes were eventually selected, based on several experimental criteria: 1) documented activity of subterranean termites within 1 meter of the structure, 2) no liquid termiticide application within the previous 5 years, and 3) no significant structural infestations detected during the initial inspection. Participating homes were in Alameda, Contra Costa, Los Angeles, Orange, and Santa Clara counties. Bait stations, baits, service equipment, and, in some cases, training, were provided by manufacturers.
The UC research team and the PCOs installed bait systems according to product labels, usually with one bait station for every 10–20 linear feet of the structural perimeter. Since all 15 sites had confirmed termite activity at the perimeter, all stations installed contained active bait, rather than monitors. The UC research team installed monitoring stations with wooden blocks immediately adjacent to each bait station. The UC team then visited each participating home every 3 months for 2 years, checking termite activity within monitoring stations and collecting termites whenever possible. The PCOs and the UC team visited each participating home every 6 months to check termite activity within bait stations, replenish baits (as per product label), and to collect termites. Collected termite specimens were sent to a collaborating lab for DNA analysis, where each sample was assigned a “Colony ID” based on its genetic signature, distinguishing it from all other colonies. At the end of the 2-year period, a final structural inspection was conducted at each home.
Findings
Most importantly, despite significant termite pressure, none of the 15 homes became infested during the study period. Foraging termites were observed and collected during initial inspections, from wood blocks during quarterly inspections, and from bait matrices during bi-annual inspections with PCOs. In some cases, termites were observed and collected from bait stations only 6 months after installation. 132 separate samples of western subterranean termites (Reticulitermes hesperus species complex) were collected. DNA analysis revealed that many of our research sites included between 3 and 5 unique colonies; 1 property included 15 unique colonies! Bait was consumed at all sites, to varying degrees. No termite colony recovered from bait stations was ever detected again.
These observations strongly suggest that all three studied bait systems were effective at eliminating termite colonies and at preventing structural infestations over a 2-year period. Furthermore, post-project surveys conducted with property owners and PCOs indicated that all parties were satisfied with the services provided and control achieved; several companies new to baiting have now embraced the program we demonstrated as a new service offering for their customers.
Reducing “time-to-attack”
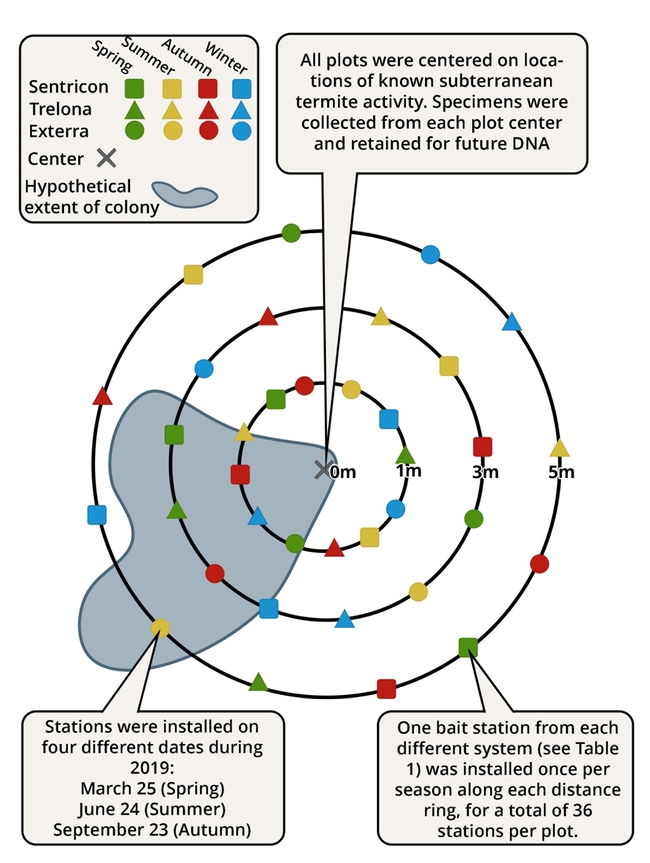
Our second objective in this research project was to investigate factors influencing bait interception time (also called “time-to-attack”). One explanation for lengthy bait interception times in California may be the interaction of climate (hot summers with little to no rain) and soil texture (high proportions of clay). Termite foraging at or near the soil surface may be limited or even nonexistent during summer months, especially when areas are not irrigated. Some research supports this idea: western subterranean termites have been observed to forage near the surface mostly during winter months in Southern California. This suggests that baits installed in summer may sit uninvestigated for 6 months or more. To test this hypothesis, we established five research plots at the UC Berkeley Richmond Field Station directly on top of areas where naturally occurring Reticulitermes termites had been observed or collected. Around these areas, we established 3 concentric rings of bait stations at 3 distances from the center, installing 1 station from each of 3 registered systems (Table 1) along each of the 3 distance rings at the beginning of each season over 1 year, for a total of 36 bait stations per plot. We didn't want to kill the termites in these plots because that would significantly confound our data, so we used cellulose bait matrices from manufacturers that did not contain the active ingredients. We also installed monitoring stations containing wood blocks at the center of each plot and along each of the three distance rings. We then checked each station every 2 months for 2 years, recording bait consumption and termite incidence.
Of the 180 bait stations and 20 monitoring stations installed, 78 bait stations and 9 monitoring stations had been hit by the end of the 2-year project period, representing an overall hit rate of 44%. Three stations were attacked within 60 days after installation, and 10 stations were attacked within 120 days. Overall, however, the average bait interception time was 367 days, supporting the general claims of California's pest control operators that baiting may take too long for most remedial termite control jobs. There were no significant differences between the three bait systems or the three distance rings.
|
Bait System, Manufacturer |
Bait Information |
Installation Specifications (for in-ground use) |
Service Specifications |
|---|---|---|---|
|
Sentricon Always Active, Corteva Agriscience |
Recruit HD Termite Bait (EPA# 62719-608): cellulose tube, 0.5% noviflumuron |
≤ 20 feet intervals; buildings, fences, decking, utility poles, trees |
Inspections at least once annually; replace bait if damaged or ≥ 1/3 consumed |
|
Advance Termite Bait System (ATBS), BASF |
Trelona Compressed Termite Bait (EPA# 499-557): cellulose wafers in plastic housing, 0.5% novaluron |
≤ 20 feet intervals; buildings, trees, wood piles, landscape elements, railroads |
Inspections at least once annually; replace bait if damaged or ≥ ½ consumed |
|
Exterra Termite Baiting System, Ensystex |
Isopthor Termite Bait (EPA# 68850-2): cellulose wafers within burlap sachet, 0.25% diflubenzuron |
≤ 20 feet intervals; buildings and other structures |
Inspections every 45 – 120 d, up to six months allowed; replace bait “after sufficient consumption” |
Our study's main question was whether installation season significantly impacts “time-to-attack” due to seasonal differences in termite foraging in California. To answer this, we pooled data from all five sites and all three bait systems and then considered just the first year of observations. The result was clear: baits installed at the beginning of winter (December 16) were intercepted ~100 days faster than baits installed at the beginning of summer (June 24)!
Conclusions
Bait stations systems may be very useful pest control tactics for use against subterranean termites in California, especially when dealing with very large colonies of native western subterranean termites, multiple colonies, sensitive sites, or sites where liquid treatments have failed. According to the labels of the three products evaluated, systems can be installed with active ingredients present on Day 1, provided a licensed Field Representative has detected and identified the target species at the site. Licensed Applicators may, according to label language and California's Structural Pest Control Act, then service bait stations, replenishing bait that has been consumed or damaged. Two of the systems evaluated allow for annual inspections, while one allows for bi-annual (every 6 months) inspections. Operators in California may decrease the bait interception time, and therefore the perceived early efficacy, by targeting initial installations for the beginning of the wet season.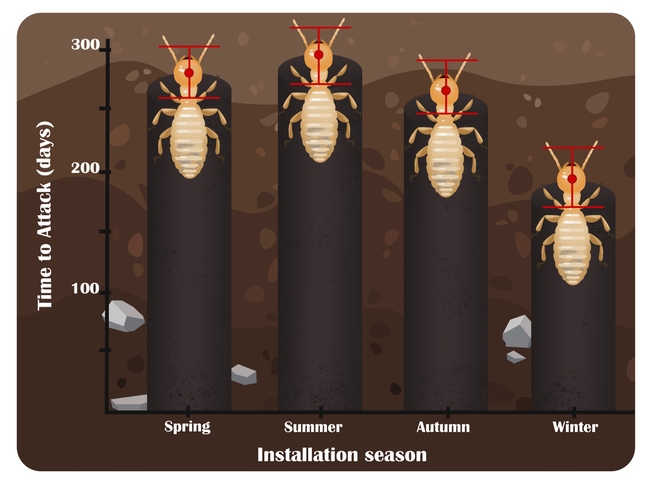
[Originally featured in the Fall 2023 edition of the Green Bulletin Newsletter for structural and landscape pest professionals.]
- Author: Belinda Messenger-Sikes

Have you been seeing a lot of defoliated sycamore trees recently? Sparse foliage and early leaf drop on sycamore trees might be due to anthracnose. The cool, wet spring in many parts of California provided the perfect conditions for this disease. Anthracnose is a common fungal disease sometimes called leaf, shoot, or twig blight. It can cause twisted, distorted branches in American sycamore, some varieties of London plane trees, and California sycamore trees. Sycamore anthracnose is primarily an aesthetic concern since it usually doesn't kill established trees.
Symptoms
Take a close look at the fallen leaves for the characteristic irregular blotches caused by this disease. Anthracnose can cause leaf spots, cankers, and wilting of young leaves in the spring (Figure 1). Emerging leaves turn brown and die. On older leaves, irregular spots or brown blotches begin on either side of the main leaf vein. The lesions spread, covering much of the leaf surface. Extensive twig or shoot blight occurs when young, growing shoots are killed. New shoots grow back from lower on the branch, eventually causing the branch to look gnarled with a “shepherd's hook” appearance (Figure 2).
Springtime disease
The fungus overwinters on infected sycamore twigs and dead leaves., The disease is most severe during wet years when temperatures during the leafing-out period are below 55°F. The disease is not a problem in hot, dry weather. While the first flush of leaves might be lost to the disease, the canopy will regrow as the second flush of growth matures.
Management
Sycamore anthracnose is best managed by cleaning up fallen infected leaves to prevent the spread of the spores to the following year's new growth. Rake and dispose of fallen leaves and twigs during the growing season and in the fall. Prune and dispose of infected twigs and branches in the fall or winter. Avoid sprinkler irrigation that keeps trees wet. When planting sycamore trees in gardens and landscapes, choose resistant cultivars.
Fungicides do not reliably control sycamore anthracnose in California. By the time you see symptoms, it's too late to treat since fungicides effective against the disease are preventive (before the symptoms appear), not eradicative (after the disease symptoms appear). In landscape and residential settings, chemical spray applications are usually not practical due to the size of the trees and the high potential for pesticide drift.
Most sycamore trees will fully recover from defoliation caused by anthracnose, so the best option could be to just rake up the fallen leaves to reduce disease inoculum for the following year.
For more information about anthracnose, see the UC IPM Pest Notes: Anthracnose.
[Originally published in the Summer 2023 Issue of the UC IPM Green Bulletin Newsletter]
If your company does residential landscape pest control, your employees should be trained to know what to do when they encounter a vegetable garden, or fruit or nut trees in a yard (Figure 1). Their training should include how to answer a customer's questions about the safety of their pesticides around vegetables or herbs. Talking to a customer about the edible plants in their garden so you don't accidentally spray their plants might save that account from subsequent cancellation.

Is it appropriate for a technician to recommend that a resident simply wash their vegetables after having their yard treated for say, mosquitoes, or should the vegetables be thrown away? What about a perimeter spray around the home? The answer to these questions depends on whether the plants were directly exposed to the sprayed pesticide and what the product label says about proximity to edible crops.
Insecticides used for perimeter sprays and mosquito control generally don't allow application to edible plants. For example, the Suspend Polyzone label states “do not apply this product to edible crops.” The Termidor SC label says, “DO NOT treat within a distance of one foot out from the drip line of edible plants. DO NOT treat fruit-bearing or nut-bearing trees.” Some product labels make no mention of vegetables or edible crops at all. If application on edible plants is not explicitly mentioned, it is not allowed.
Will pesticides make a plant toxic?
Many insecticides, including some common active ingredients familiar to urban pesticide applicators, are used legally on agricultural crops. This is allowed by the United States Environmental Protection Agency (US EPA) only if that pesticide has been granted a tolerance for a given crop, and certain days-to-harvest intervals are followed. These rules work to ensure that any pesticide residues left after a pesticide application are below levels of concern for human health. The 2019 Pesticide Data Program survey by USDA shows that this system works. Out of 10,000 market food samples analyzed in the study, nearly 99% had residues well below the EPA established tolerances. More than 42% had no detectable pesticide residue.
Insecticide residues on plants are not necessarily acutely toxic, especially when label directions are followed, and adequate time passes to allow the product to naturally degrade. The products used by professional pesticide applicators may contain the same active ingredients used by farmers; but they may differ in concentration and formulation. Most importantly, insecticides used for landscapes and homes do not carry food-treatment labels, so they cannot legally be used on edible crops.
Talstar products, for example, consist of the active ingredient bifenthrin, the same active ingredient used by farmers and even home gardeners under a variety of trade names. The Talstar® P label however, says "do not use on edible crops." You must follow the label for the product you are using.
Spray contamination
If an insecticide is deliberately sprayed on an edible crop or plant, and the product is not labeled for such use, the plant would not be considered safe by EPA standards. The implication is that the whole plant, or at least the edible parts, should be thrown away. Your customer could replant unless that is prohibited by the label.
Backpack mistblowers are commonly used for applying residual insecticides in areas such as mosquito resting sites; but mists should be applied carefully to avoid drift onto fruit and nut trees and vegetable gardens.
Labels generally do not, however, prohibit use of these products in the vicinity of a vegetable garden. Take care to keep sprays directed away from vegetable gardens, so that any incidental drift from nearby spraying does not land on edible plants. Thermal foggers and ULV applications used nearby should leave insignificant residues if the application orifices are always directed away from edible plants.
Applicators should always be aware of weather conditions and the locations of edible plants. If wind is blowing toward a garden, upwind applications should be avoided.
What should you do if a fruit, nut, vegetable, or herb is accidentally sprayed? Notify the customer that an accidental spray contamination has occurred. Annual plants should be pulled. For both annual and perennial plants, the produce must be discarded by the customer and not eaten.
Systemic insecticides
Some insecticides are "systemic," meaning they have enough water solubility to be taken up by plant roots and translocated to other parts of the plant. Although the EPA allows some systemic insecticides on crops, in general systemics are not labeled for use on food crops because they can leave residues in edible plant tissues that do not quickly degrade.
Insecticides containing neonicotinoids and acephate are examples of professional grade insecticides that may be systemic in plants. These include products like Premise, Alpine, Tandem, Transport, Temprid, Orthene and others. Herbs and other root or leafy vegetables exposed to systemic insecticides should be considered contaminated for the season and should not be consumed.
Some termiticides can also be systemic in plants, leading to concerns about vegetable gardens and fruit trees planted next to homes treated for termites. Fipronil, for example, is slightly systemic in some plants; and the Termidor SC label says not to apply around edible plants. The label does not say explicitly how far away an edible plant must be, although the Premise Pro label (whose active ingredient, imidacloprid, is much more water soluble) is more specific. It says to "not treat within a distance of one foot out from the drip line of edible plants." The Premise instruction is probably a good, conservative guidelines for all termiticides. Keep the outermost leaves of garden plants at least a foot away from any soil-applied termiticide.
Washing Crops
A concerned customer whose nearby yard or house perimeter has been treated with an insecticide spray should consider washing their vegetables or other harvested edibles. Washing is a good idea whether p
esticides have been used or not. The best washing technique is to gently rub off any dirt while holding the produce under running water. This is a great way to remove dust, microorganisms, and any traces of pesticides from vegetable and fruit surfaces.
Would you recognize an edible plant?

Can you and your technicians and applicators tell basil from begonia, mint from marigold, or pear from poplar? Applicators following label directions around a home needs to be aware of what plants are present. You don't have to be a botanist or know all the local tree species; but you should recognize the most common fruit and nut trees, herbs, and vegetables (Figure 2).
When visiting a residence for the first time, ask your customer if they have any herbs, fruit trees, nut trees, or vegetables. Gardeners may plant edible plants within flower gardens, so they might have a basil plant or a tomato plant growing among the daisies. Assume your customers don't use pesticides in their vegetable garden and avoid these areas accordingly.
Your customers will appreciate any extra consideration you give to their edible garden plants. Treat them well and they might even greet you at the door with a big bag of zucchini!
Based on the article “Bugs and basil: Insecticides and veggies don't always mix” by Dr. Mike Merchant, Texas AgriLife Extension at https://insectsinthecity.blogspot.com/2018/08/pesticides-and-veggie-gardens.html
[Originally featured in the Fall 2021 edition of the Green Bulletin Newsletter for structural and landscape pest professionals.]
- Author: Beatriz Nobua-Behrmann
- Author: Randall Oliver
- Posted by: Elaine Lander
Two identical looking species of wood-boring beetles, collectively known as invasive shothole borers (ISHB), have killed thousands of trees in Southern California and pose an ongoing threat to California's urban and wildland forests. These beetles, which are not native to the United States, were first identified in Los Angeles County in 2012 and have since spread to six other counties: Orange, Riverside, San Bernardino, San Diego, Santa Barbara, and Ventura.
Beetles, Fungus, and Impact


As the fungus grows, it colonizes the tree's vascular system, blocking transport of water and nutrients. This causes a disease called Fusarium dieback that can kill branches or entire trees. The beetles and their symbiotic fungi have a wide range of suitable hosts, including more than 65 species of trees found in California. The most highly susceptible trees include many of the species commonly used for landscaping, such as sycamores, oaks, cottonwoods, and box elder trees. ISHB beetles attack healthy trees as well as stressed or diseased trees in a variety of urban, suburban, and riparian settings. Visit www.ishb.org to find the full list of reproductive hosts in California.
Female beetles can fly short distances, allowing the pest-disease complex to spread into new areas near already infested trees. Beetles can also be transported in infested firewood and green waste, leading to spread over much greater distances. While beetles have only been identified in Southern California and the Central Coast to date, further spread throughout much of California is possible.
ISHB-infested trees can quickly become public safety hazards. Trees with heavily infested branches can be especially hazardous, since the combined damage of the fungal disease and the beetle's tunneling activity weakens the wood, causing limbs to break and fall. In addition, severely infested trees will become constant sources of beetles that can disperse and infest neighboring trees. Such “amplifier” trees generally need to be removed completely, while more lightly infested trees can be managed or treated without requiring removal. Therefore, early detection and rapid response is the key to controlling ISHB. Substantial recovery in lightly to moderately infested trees has been observed in some areas where amplifier trees have been removed.
Identifying an ISHB infestation

- Beetle entry holes: When the beetles tunnel into trees they make small, perfectly round holes, each about the size of the tip of a medium ballpoint pen (0.8 mm). (Figure 3)
- Additional signs and symptoms: Entry holes are usually accompanied by one or more of the following signs and symptoms, which vary by the tree species: staining, gumming, white powdery exudate, or frass (boring dust).
- Dieback: Dead or wilting branches can be a sign of a severe infestation. If you see dieback on trees, check for entry holes on the trunk or the branch collars.
In addition to visual inspections, traps using quercivorol, a plant-based lure that attracts the beetles over short distances, can help determine the presence and abundance of beetles in an area. Trapping is especially useful for large or inaccessible areas where regular visual inspections of all the trees are not practical. In those cases, trapping can help determine if ISHB is present in the area and can help focus survey efforts on infested trees. Because the lure has relatively low attractiveness, trapping is not an effective control method for ISHB and is only used for detection purposes.
What Can You Do?
- Keep trees healthy. Proper irrigation and maintenance will keep trees strong and help protect them from ISHB and other pests.
- Prune out infested branches. Removing branches that have clusters of 50 ot more ISHB holes would help control this pest. For trees that undergo heavy pruning every year, like avocado trees, removal of all infested branches is recommended. Tools should be disinfected after pruning by spraying them with a solution of 5% disinfecting bleach or 70% ethanol to avoid spreading the fungal disease to other trees.
- Remove severely infested trees. Unfortunately, severely infested trees (with more than 150 entry holes and ISHB-related dieback) are not likely to survive. These trees should be removed as soon as possible, and the stump should be ground to one inch or less. (Figure 4)
- Manage downed wood. Green waste generated by branch and tree removals should be properly disposed.
- Chipping/grinding to one inch or less kills 99.9% of the beetles. If that is not possible, chipping to three inches or less still will kill 98% of the beetles in the wood. In already infested areas, chipped wood can be used onsite as mulch. However, if working on a newly infested area or if the wood chips will be moved to another area, chipping should be combined with solarization or composting to kill 100% of the beetles. If chipping is not an option, logs can also be solarized or kiln dried to exterminate the beetles.
 Figure 4. California sycamore heavily infested with invasive shothole borers. (Credit: B Nobua-Behrmann, UCCE)
Figure 4. California sycamore heavily infested with invasive shothole borers. (Credit: B Nobua-Behrmann, UCCE) - Solarization involves covering the material with clear plastic tarp and letting the heat from the sun kill the remaining beetles. Chips and beetles should be fully contained by wrapping plastic both underneath and over the material. Chips should remain covered for at least six weeks during the summer months or for at least six months between September and June. The depth of the pile should be no more than 30 inches deep, to ensure even heating.
- Composting, when done correctly, should also kill the remaining beetles in the chips. It is recommended to send infested chips to a professional composting facility that has earned the U.S. Composting Council's Seal of Testing Assurance.
- Prevent the spread. Avoid spreading this pest by not moving firewood or mulch that hasn't been properly solarized or composted. If you must move infested greenwaste (for example, to bring it to a composting facility) make sure the load is tightly covered while in transit.
- Consider chemical control.TreesthatarereproductivehostsforISHB and that show signs of active infestations can be treated with a combination of insecticide and fungicide. The decision to treat a particular tree depends, among other things, on the tree's condition, value, and hazard level. Trees that aren't already infested should be monitored but not treated. There are various chemicaloptionsthatcanbeusedagainstISHB-FD.
- Trunk sprays of a contact insecticide, such as bifenthrin, combined with Bacillus subtilis or tebuconazole (which are fungicides) have been demonstrated to offer some degree of control.
- Systemic soil injections or drenching with the insecticide imidacloprid has also provided control, as has trunk injection with emamectin benzoate (insecticide) combined with tebuconazole or propiconazole (fungicides).
- These pesticides should only be applied by a licenced professional following the instructions on their labels to avoid harming non-target organisms.
Biocontrol options are currently under research. They include the use of natural enemies (such as parasitic wasps from the beetles' point of origin), entomopathogenic fungi (which are fungi that attack insects), endophytes (which are microorganisms that live in the tree that may provide protection) and nematodes. But these biological management options might take time before they are tested and available. Until then, prevention, early detection, and rapid response are our best weapons to keep trees healthy and alive. For more information on invasive shothole borers and their management, visit www.ishb.org.
[Article originally published in the Summer 2021 issue of the Green Bulletin.]